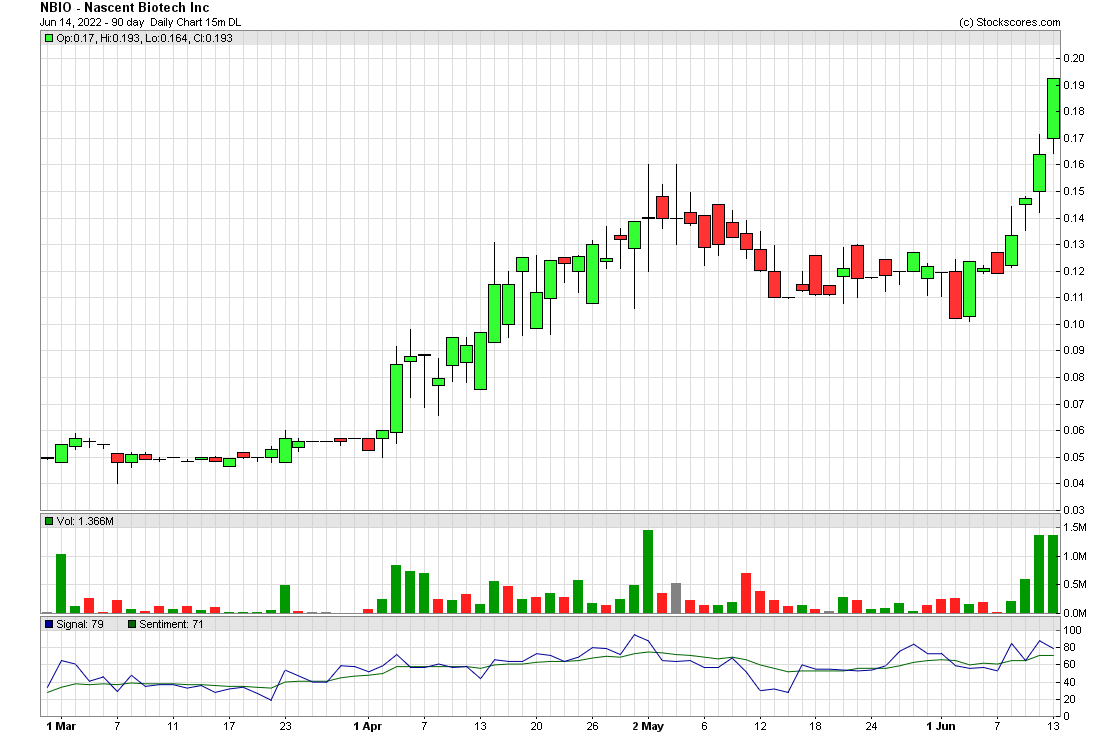
Nascent Biotech Inc (OTCMKTS: NBIO) is making a steady run northbound off its base at a nickel and saw some of its biggest gains while the overall markets saw steep declines. NBIO is a low float biotech whose lead product Pritumumab (PTB) targeting Glioblastoma is being studied in Phase I clinical trials. On Friday the Company announced the completion of the Third cohort in dosing patients for its Phase I trial for Brain Cancer. After reviewing the data gathered from the first three cohorts, this milestone will allow the trial to advance to the fourth and possibly final cohort.
Pritumumab works by binding to a target on the surface of cancer cells called ectodomain vimentin (also referred to as cell-surface vimentin). The target, generally referred to as an antigen, is prevalent in many different tumor types and is not being targeted by any other biopharmaceutical companies. By binding to this target, Pritumumab is able to make the tumor cells “known” to the body’s immune system, resulting in potentially several types of immune responses, including anti-idiotype, apoptosis, antibody-dependent cellular cytotoxicity and complement-dependent cytotoxicity, leading to death of the cancer cells and overall depletion of the tumor.
Nascent Biotech Inc (OTCMKTS: NBIO) operating out of Vero Beach, Florida is a clinical-stage biotech company pioneering the development of monoclonal antibodies to be used in the treatment of various cancers and viral infections, helping people worldwide. Its products are not yet commercially available. The Company’s lead candidate, Pritumumab (PTB), is a monoclonal Antibody (Mab) that the Company is developing as a treatment for epithelial cancers (which includes lung, breast, colon, brain, and pancreas). Currently Pritumumab (PTB) has received FDA orphan drug status and is being studied in Phase I clinical trials for the treatment of Brain Cancer. The Company is led by CEO Sean Carrick who has an extensive track record of success with a career that spans more than 30 years of building and leading successful Life Science Companies in large, mid-cap and venture-backed stages
Current brain cancer therapeutic strategies include chemotherapy drug Temodar®, surgery, and/or radiation. Even when removed, most brain tumors come back within one-year post-surgery. With current standards of care, only 58% of brain cancer patients live past the first year after diagnosis with certain types, anaplastic astrocytoma and glioblastoma, the five-year survival rates are 27% and 5%, respectively
Based on pre-clinical and clinical studies, the Company believes Pritumumab may offer an advantage over existing treatments. Nascent has addressed manufacturing questions by re-engineering antibody production into the commonly used CHO cell expression system. Pritumumab works by binding to a target on the surface of cancer cells called ectodomain vimentin (also referred to as cell-surface vimentin). The target, generally referred to as an antigen, is prevalent in many different tumor types and is not being targeted by any other biopharmaceutical companies. By binding to this target, Pritumumab is able to make the tumor cells “known” to the body’s immune system, resulting in potentially several types of immune responses, including anti-idiotype, apoptosis, antibody-dependent cellular cytotoxicity and complement-dependent cytotoxicity, leading to death of the cancer cells and overall depletion of the tumor.
$NBIO just keeps on moving up in this terrible market. Only the strong will shine through!! 💪🏼💪🏼 @Nascent_BioTech
— Wall Street Money (@wallstreetmon) June 10, 2022
Nascent expects to amplify on the past clinical development strategy during the next 12 months plan to: complete Phase I clinical trials in the United States, establishing safety and the optimal dose of Pritumumab for treatment of patients with brain cancers. Commence Phase 2 clinical trials in the United States and China with Pritumumab for the treatment of lung cancer and breast cancer that has metastasized to the brain and continue to evaluate the application of Pritumumab in the treatment of other ectodomain vimentin positive cancer types (pancreatic, lung and colon) where there may be unmet medical needs.
On December 7, 2018, Nascent received FDA clearance to begin clinical trials with its first drug product lot but left the Company on partial clinical hold for the bulk substance lot. On December 1, 2020, the Company received FDA clearance to begin clinical trials with the second lot. The Company commenced the Phase I trial. In March 2021, the Company enrolled its initial patient. In May 2021, the first three patient cohort was enrolled.
On June 7 NBIO announced the completion of the Third cohort in dosing patients for its Phase I trial for Brain Cancer. After reviewing the data gathered from the first three cohorts, this milestone will allow the trial to advance to the fourth and possibly final cohort. As the fourth cohort is underway, Nascent will also be performing PK studies to help determine if a fifth cohort is needed. Patient enrollment will continue for Phase I. Anyone interested may review trial requirements at www.clinicaltrials.gov, then search Pritumumab.
Nascent CEO Sean Carrick announced “Having completed the first three cohorts, we are excited to move to our fourth and possibly final cohort in the Phase I trial. The third cohort took longer than expected but the data received is very useful moving forward,” noted Nascent CEO, Sean Carrick, further stating that,
According to a recent ResearchAndMarkets report the global brain tumor drugs market is expected to grow from $3.17 billion in 2021 to $3.49 billion in 2022 at a compound annual growth rate (CAGR) of 10.2%. The growth is mainly due to the companies rearranging their operations and recovering from the COVID-19 impact, which had earlier led to restrictive containment measures involving social distancing, remote working, and the closure of commercial activities that resulted in operational challenges. The market is expected to reach $4.84 billion in 2026 at a CAGR of 8.5%.
$NBIO News June 07, 2022
PTB is used as a targeted immunotherapy unlike chemotherapy targets only cancer cells without damaging healthy cells.https://t.co/P14ltZnZjf
— papaspoppinpennys (@papaspoppinpeny) June 7, 2022
Currently trading at an $18 million market valuation, NBIO has 113,063,175 shares outstanding, 57,804,675 of which are restricted leaving 55,258,500 free trading NBIO shares worth $9 million. NBIO is fully reporting OTCQB and recent filings show the entire NBIO management team acquiring and exercising stock options at $0.05 in April. Since than the stock has been rising steadily as the Pritumumab (PTB) Phase I trials progress. Pritumumab targets the Glioblastoma market worth $3.5 billion globally. On Friday the Company announced the completion of the Third cohort in dosing patients for its Phase I trial. After reviewing the data gathered from the first three cohorts, this milestone will allow the trial to advance to the fourth and possibly final cohort. The Company hopes to progress to Phase 2 clinical trials in the United States and China with Pritumumab for the treatment of lung cancer and breast cancer that has metastasized to the brain and continue to evaluate the application of Pritumumab in the treatment of other ectodomain vimentin positive cancer types (pancreatic, lung and colon).